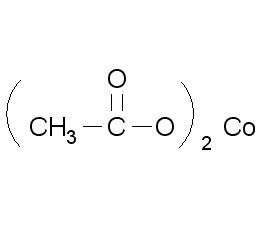
Structural formula
| Business number | 01GK |
|---|---|
| Molecular formula | C4H6CoO4 |
| Molecular weight | 177.02 |
| label |
Cobaltous oxalate, Cobaltous oxalate, cobalt acetate, Cobalt acetate tetrahydrate, Cobaltousdiacetate, catalyst, Desiccant for paints and coatings, printing and dyeing mordant, FRP curing accelerator, Hidden ink |
Numbering system
CAS number:71-48-7
MDL number:MFCD00008689
EINECS number:200-755-8
RTECS number:AG3150000
BRN number:None
PubChem number:24864856
Physical property data
1. Properties: purple-red crystals that are easily deliquescent. Slightly acetic acid smell, easy to deliquesce
2. Density (g/mL, 25/4℃): 1.7043
3. Relative vapor density (g/mL, air=1) : Uncertain
4. Melting point (ºC): 290
5. Boiling point (ºC, normal pressure): Uncertain
6. Boiling point (ºC, 5.2 kPa): Uncertain
7. Refractive index: Uncertain
8. Flash point (ºC): Uncertain
9. Specific rotation ( º): Uncertain
10. Autoignition point or ignition temperature (ºC): Uncertain
11. Vapor pressure (kPa, 25 ºC): Uncertain
12. Saturated vapor pressure (kPa, 60 ºC): Uncertain
13. Heat of combustion (KJ/mol): Uncertain
14. Critical temperature (ºC) : Uncertain
15. Critical pressure (KPa): Uncertain
16. Log value of oil-water (octanol/water) partition coefficient: Uncertain
17. Explosion upper limit (%, V/V): Uncertain
18. Explosion lower limit (%, V/V): Uncertain
19. Solubility: Dissolved in Water, acid and ethanol, insoluble in acetone and benzene,
Toxicological data
Acute toxicity: Rat oral LD50: 503 mg/kg; Mouse intravenous LD50: 31 mg/kg; Rabbit intravenous LD50: 25 mg/kg;
Mutagenicity: Morphological transformation of hamster embryos Test system: 200 umol/L;
Ecological data
None
Molecular structure data
None
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 4
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 80.3
7. Number of heavy atoms: 9
8. Surface charge: 0
9. Complexity: 25.5
p>
10. Number of isotope atoms: 0
11. Number of determined atomic stereocenters: 0
12. Number of uncertain atomic stereocenters: 0
13. Determine the number of stereocenters of chemical bonds: 0
14. Uncertain number of stereocenters of chemical bonds: 0
15. Number of covalent bond units: 3
Properties and stability
1. Loss of crystal water above 140°C. Easily soluble in water and ethanol, the solubility in 100g ethanol at 15°C is 1.49g. Soluble in acetic acid and pyridine, insoluble in acetic anhydride, insoluble in acetone and benzene. Toxic and carcinogen. Should be kept closed.
2.Harmful to the body if inhaled or ingested. Inhalation can cause pharyngitis, followed by gastrointestinal irritation symptoms, such as vomiting, abdominal cramps, increased body temperature, and calf weakness. Skin contact can cause allergic dermatitis and contact dermatitis.
Storage method
1. This product should be sealed and stored in a cool, dry and dark place. Store in a cool, dry place away from sunlight and rain.
2.Should be stored separately from moist items, edible chemical raw materials, etc.
Synthesis method
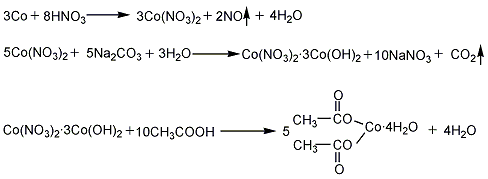
(1) Metal cobalt or cobalt oxide method First, metal cobalt or cobalt oxide reacts with nitric acid or sulfuric acid to produce cobalt nitrate or cobalt sulfate, and then reacts with sodium carbonate or ammonium bicarbonate to generate cobalt carbonate, and then adds acetic acid. Reaction, the reaction product is evaporated, concentrated, crystallized and dried to obtain the finished product.
Each ton of product consumes 265kg of metallic cobalt (>99%), 1,000kg of acetic acid (industrial product), 1,200kg of sodium carbonate (industrial product), and 1,100kg of nitric acid (industrial product).
(2) Cobalt sulfate method will Cobalt sulfate reacts with soda ash by heating to generate cobalt carbonate, which is then reacted with acetic acid. The reaction product is filtered, concentrated, crystallized, dehydrated and packaged to obtain the product.
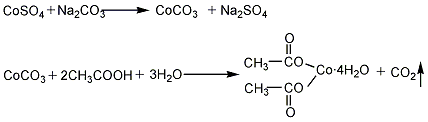
3. Take 5g of nitric acid hexahydrate Add cobalt to 25 mL of acetic anhydride, mix evenly, heat and boil for 10 minutes, and allow crystals to precipitate after standing. Filter with suction and wash with a small amount of acetic anhydride and diethyl ether. Drain. Dry under vacuum at 100°C for 1 hour.
Purpose
1. Catalyst for xylene oxidation, used in the production of desiccants for coatings, mordants for printing and dyeing, FRP curing accelerators and hidden inks, etc. 2. Used as a catalyst for transesterification reaction, the dosage in the production of polyester resin is about 001% to 005%. This product is mainly used as an oxidation catalyst for organic synthesis (such as a catalyst for producing terephthalic acid, the basic raw material of polyester resin, from the oxidation of paraxylene). It is also used to prepare desiccants for paints and coatings, mordants for printing and dyeing, FRP curing accelerators and visible inks. 3.It is used in the dyeing and sealing treatment of oxide films. In the sealing solution of aluminum and alloy anodization, appropriate cobalt salt can also be added to improve its sealing performance. . It can also be used as an additive for zinc plating and the addition of cobalt salts for electroplating cobalt alloys, electroless cobalt plating and alloys.
extended-reading:https://www.bdmaee.net/niax-ef-708-foaming-catalyst-momentive/extended-reading:https://www.bdmaee.net/nt-cat-la-210-catalyst-cas10861-07-1-newtopchem/extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/-EG-33-triethylenediamine-in-EG-solution-PC-CAT-TD-33EG.pdfextended-reading:https://www.newtopchem.com/archives/category/products/page/171extended-reading:https://www.bdmaee.net/wp-content/uploads/2021/05/144-1.jpgextended-reading:https://www.newtopchem.com/archives/1911extended-reading:https://www.cyclohexylamine.net/category/product/page/23/extended-reading:https://www.cyclohexylamine.net/catalyst-1028-polyurethane-catalyst-1028/extended-reading:https://www.newtopchem.com/archives/44782extended-reading:https://www.bdmaee.net/lupragen-dmi-polyurethane-gel-catalyst/


Comments